Question: How many moles are in 1000 g of sucrose?

1. Obtain the atomic mass
First, write down the formula for sucrose: C12H22O11

2. Calculate the molar mass of sucrose C12H22O11
The formula tells us there are 12 carbon atoms, 22 hydrogen atoms, and 11 oxygen atoms.
Look for the atomic mass of each element present in the compound from the periodic table.
Organize your calculations neatly to show how to calcualte the sum of the atomic masses of the compound’s constituent atoms.
| Element | Symbol | Atomic mass (amu) | #n atoms | Mass |
| Hydrogen | H | 1.008 | 22 | 22(1.008) = 22.176 amu |
| Carbon | C | 12.01 | 12 | 12(12.01) = 144.12 amu |
| Oxygen | O | 16.00 | 11 | 11(16.00) = 176.00 amu |
| Molecular mass of sucrose
= (22.176 + 144.12 + 176.00) = 342.296 amu |
The molar mass of sucrose is simply the molecular mass expressed as g/mol.
Molar mass of sucrose = 342.296 g/mol
3. Calculate the number of moles
Apply the formula to calculate the number of moles
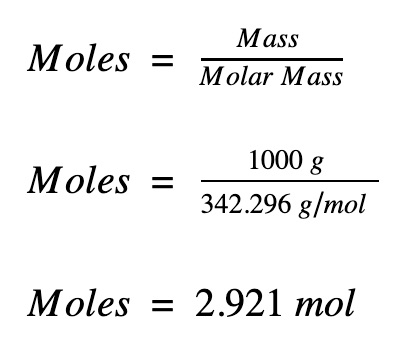
The answer can be rounded to 3 mol.
(Note: The symbol for mole is “mol”)
Usefulness of moles
- Knowing the number of moles allows you to calculate the number of atoms if you are dealing with chemical compounds.
- Moles are very useful, they allow chemists to use chemical equations to determine the mass of reactants and products.
Read about Avogadro Constant

